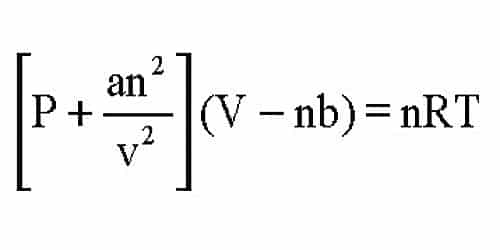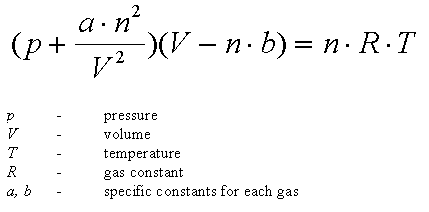Derive the values of critical constants from the Van der Waals constants? - Sarthaks eConnect | Largest Online Education Community
What are the symbols 'R' (ideal gas constant), 'a' and 'b' (Van der Waals constants) abbreviations for? - Quora

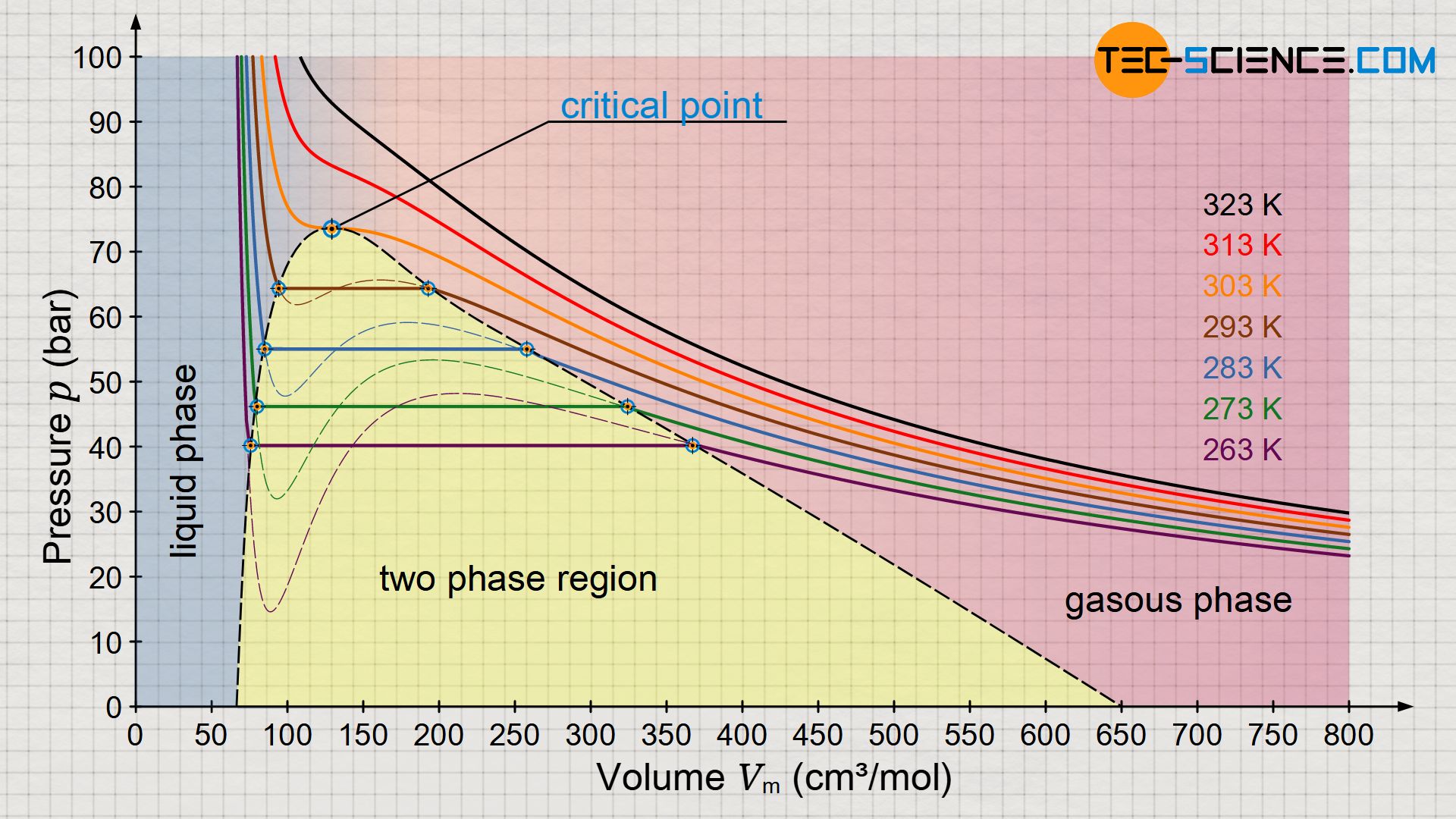
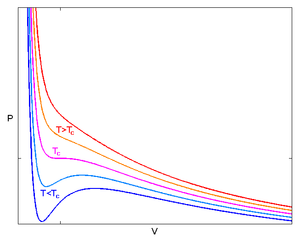
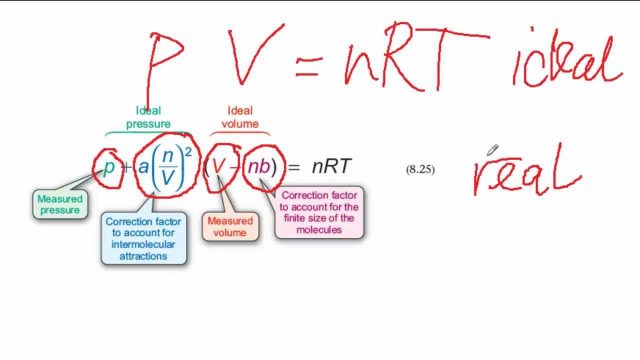
The Van der Wall equation for 1 mole of a real gas is ( P + a/V^2 )(V - b) = RT where P is the pressure, V is the volume, T