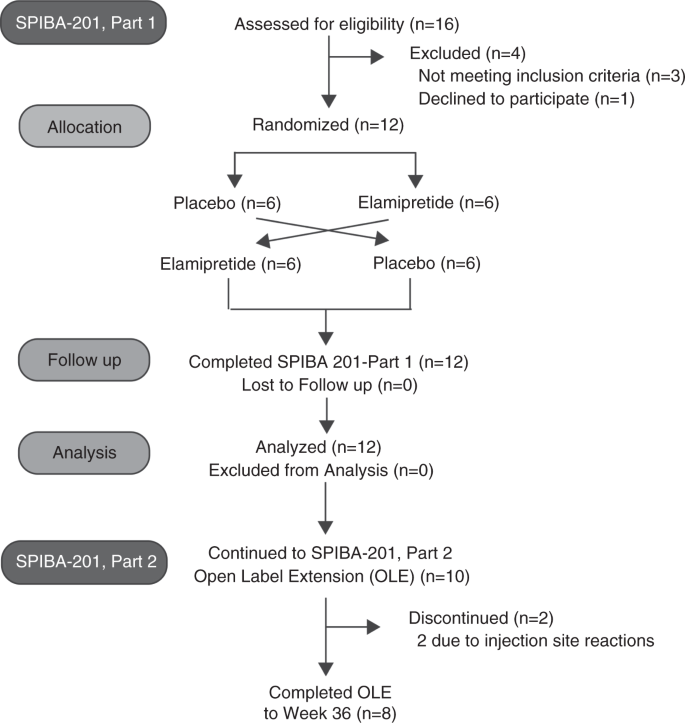
Phase II single arm open label multicentre clinical trial to evaluate the efficacy and side effects of a combination of gefitinib and methotrexate to treat tubal ectopic pregnancies (GEM II): study protocol
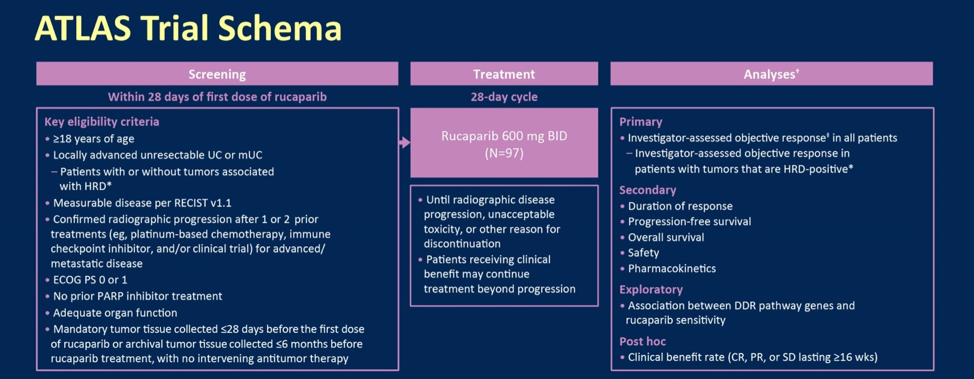
ASCO GU 2020: Results from ATLAS, a Phase 2, Open-Label Trial: Rucaparib for Recurrent, Locally Advanced or Metastatic Urothelial Carcinoma

PIONEER Trial: An OPen-label, Randomized, Controlled, Multicenter Study ExplorIng TwO TreatmeNt StratEgiEs of Rivaroxaban and a Dose-Adjusted Oral Vitamin K Antagonist Treatment Strategy | tctmd.com
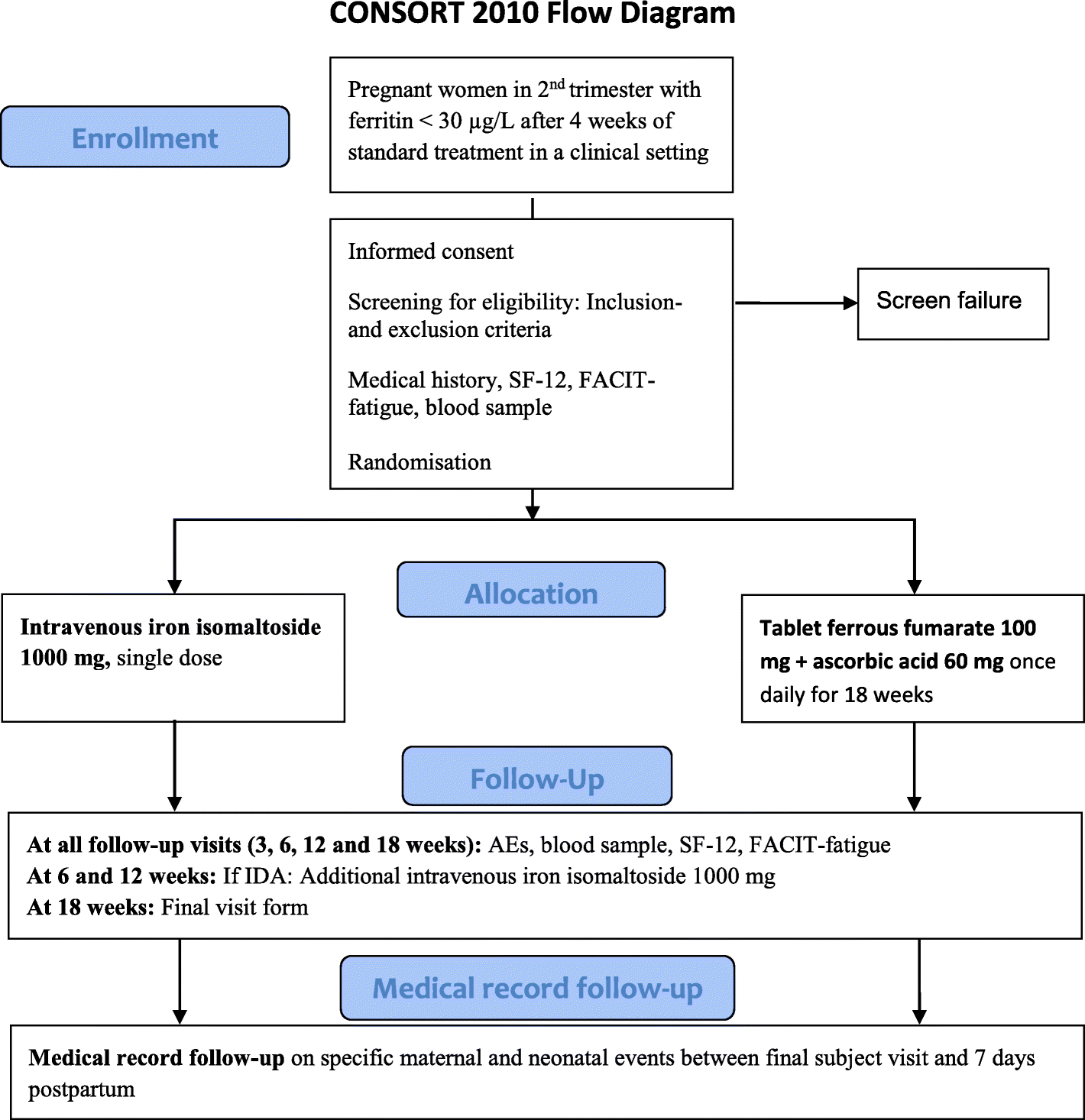
Intravenous iron isomaltoside versus oral iron supplementation for treatment of iron deficiency in pregnancy: protocol for a randomised, comparative, open-label trial | Trials | Full Text

Drug trials Learning Outcome: DESCRIBE THE USE OF OPEN LABEL, BLIND AND DOUBLE BLIND TESTS IN HUMAN TRIALS (HIGHER) AND THE USE OF PLACEBOS. - ppt download

Headache Journal on Twitter: "Transcranial Magnetic Stimulation for #Migraine Prevention in #Adolescents: A Pilot Open-Label Study #TMS SL Irwin et al @aagelfand @petergoadsby https://t.co/ThCNhNnWbu https://t.co/eummTQHnsY" / Twitter

Phase I/II open-label trial of intravenous allogeneic mesenchymal stromal cell therapy in adults with recessive dystrophic epidermolysis bullosa - Journal of the American Academy of Dermatology
![PDF] Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition. | Semantic Scholar PDF] Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/814ad6ccbfa9defca4d2b00c4672f9070cf6b8da/16-Figure1-1.png)
PDF] Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition. | Semantic Scholar
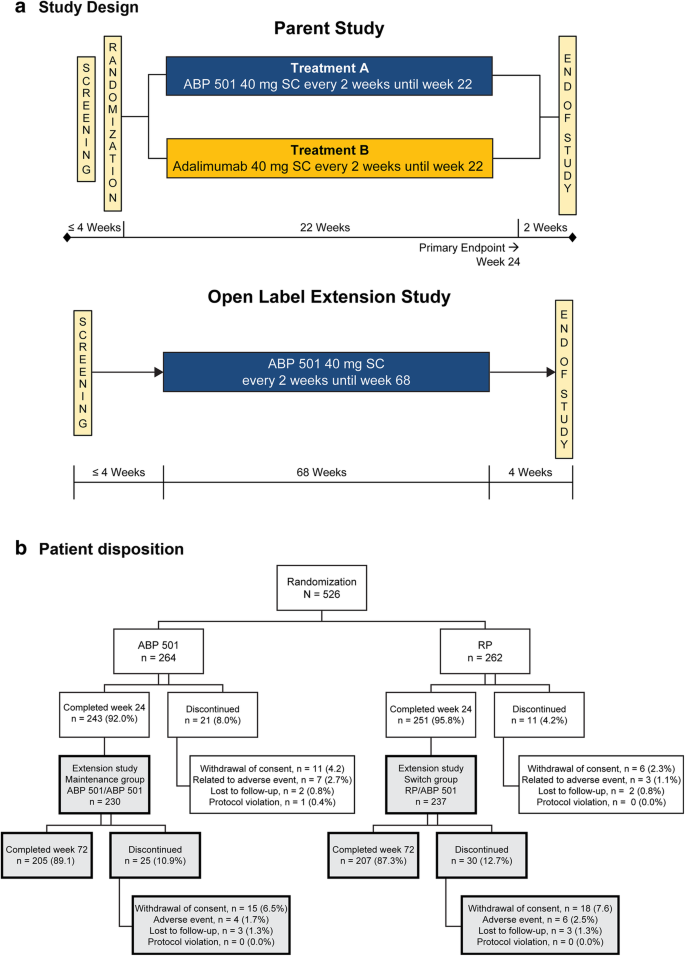
An open-label extension study to demonstrate long-term safety and efficacy of ABP 501 in patients with rheumatoid arthritis | Arthritis Research & Therapy | Full Text

Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition - Journal of Clinical Epidemiology

Safety and tolerability of adjunctive lacosamide in a pediatric population with focal seizures – An open-label trial - Seizure - European Journal of Epilepsy