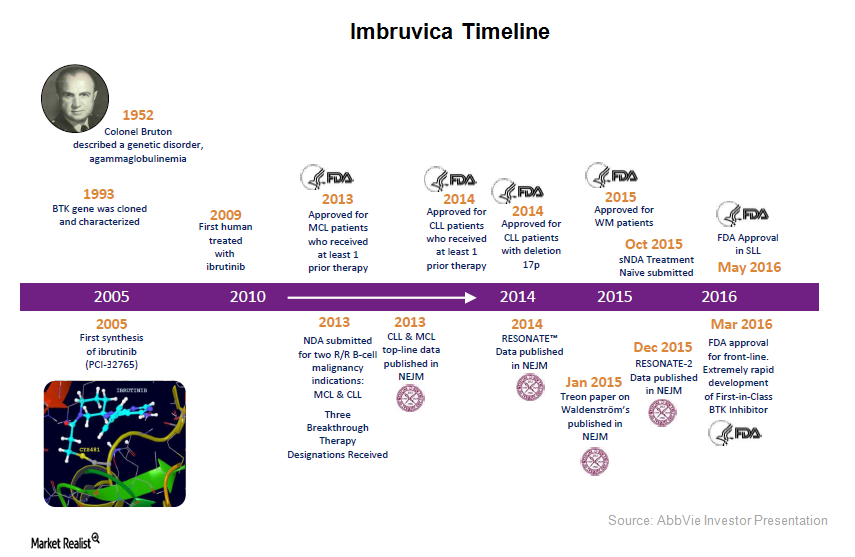
Janssen Seeks Expanded Use of IMBRUVICA® (ibrutinib) in Combination with Rituximab for Patients with Previously Untreated Chronic Lymphocytic Leukaemia (CLL) | Business Wire

These highlights do not include all the information needed to use IMBRUVICA safely and effectively. See full prescribing information for IMBRUVICA. IMBRUVICA ® (ibrutinib) capsules, for oral use IMBRUVICA ® (ibrutinib) tablets,

Supplemental Materials for Ibrutinib for patients with relapsed or refractory chronic lymphocytic leukaemia with 17p deletion (RESONATE-17): a phase 2, open-label, multicentre study - The Lancet Oncology

These highlights do not include all the information needed to use IMBRUVICA safely and effectively. See full prescribing information for IMBRUVICA. IMBRUVICA ® (ibrutinib) capsules, for oral use IMBRUVICA ® (ibrutinib) tablets,

BeiGene's Phase III Waldenström's study misses mark, but with a possible silver lining - MedCity News

BeiGene nabs landmark FDA nod for Brukinsa, kicking off challenge against blockbuster Imbruvica | FiercePharma

These highlights do not include all the information needed to use IMBRUVICA safely and effectively. See full prescribing information for IMBRUVICA. IMBRUVICA ® (ibrutinib) capsules, for oral use IMBRUVICA ® (ibrutinib) tablets,

Venetoclax for chronic lymphocytic leukaemia progressing after ibrutinib: an interim analysis of a multicentre, open-label, phase 2 trial - The Lancet Oncology